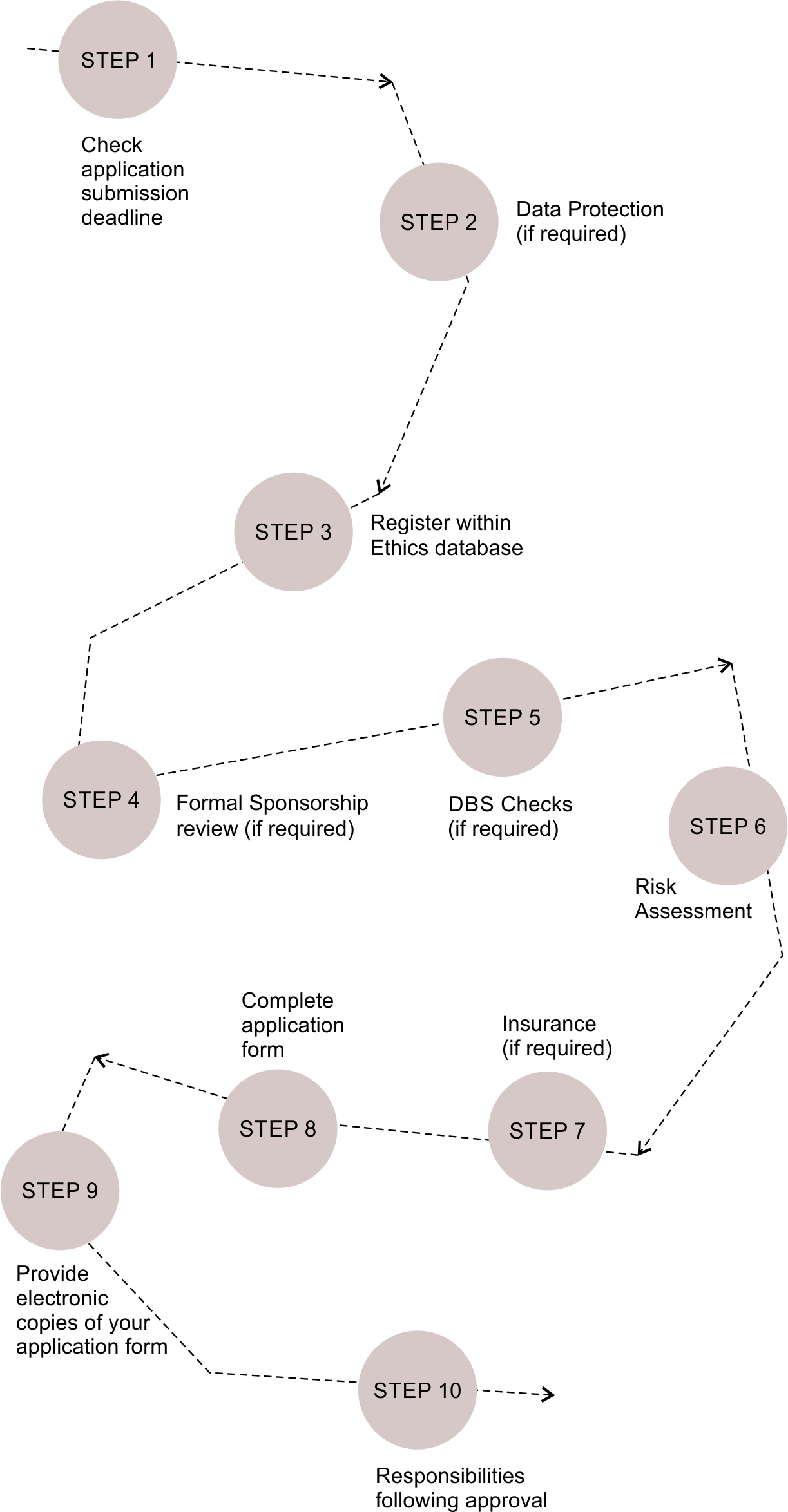
Procedures for Applications
Step 1
Check Research Ethics Committee Meeting Dates and Submission Deadlines for:
- Data Protection Registration Submission (if required);
- Ethics Applications for full committee review. Chair's review applications are dealt with on an ad hoc basis so there are no submission deadlines.
Application Deadlines & Meeting Dates →
Step 2
Data Protection (if required)
If you are proposing to collect personal data i.e. data from which a living individual can be identified you must be registered with the UCL Data Protection Officer before you submit your ethics application for review. If the Data Protection Officer advises you to make changes to the way in which you propose to collect and store the data this should be reflected in your ethics application form.
You can register for Data Protection on the → UCL Legal Services website by downloading the ‘Research Registration Form’. The form should be completed and e-mailed to: data-protection@ucl.ac.uk. The Legal Services should be able to process your application within 10 days upon which time you will be issued with a Data Protection Registration Number.
Please quote your Data Protection registration number in the appropriate section of your application form as evidence that the project has been registered with the UCL Data Protection Office.
To Note: If the Data Protection registration process is taking longer than the advertised 10 days, please go ahead and submit your ethics application form marking the sections relating to Data Protection registration as ‘to follow’ or ‘registration in progress’ and supply the ethics team with your DP registration number (together with any changes the Data Protection Office advised you to make to the way in which you propose to store and collect your data for example) once it is available so that we can tie up that information with your ethics application. However, please note that data collection cannot commence until you have received both, Data Protection and ethical approval.
Step 3
Register within the Ethics database and download an ethics application form
- You will need to register your personal (if this is your first application) details on the ethics database ( →Registration Form) and a personal account will be established for you.
- For existing users log into your account using your email address and password and add a ‘new project’ to your account.
- A unique Project ID for your proposal will be issued immediately which you should make a note of. The finance department will require this Project ID in order to process grant applications based on this research.
- Once you have entered your personal and project details, a zip folder will appear containing a ‘low’ and ‘high’ risk application form together with supporting guidelines for you to download to your computer. You will need to decide based on the criteria of minimal risk e.g. if your research study does not involve vulnerable groups, intrusive interventions [including MRI], sensitive topics and deception, it is likely that a ‘low’ risk application would be appropriate for the REC Chair's review. For studies involving more than minimal risk a ‘high risk’ application form will need to be completed for review by the full ethics committee.
- Low risk applications are reviewed by the REC Chair on an ad hoc basis so you can submit your application at any time, whereas there are application submission deadlines for high risk applications as outlined in Step 1.
Step 4
Formal Sponsorship Review for Clinical Trials Conducted in Developing Countries (if required)
If your project is an interventional study in a developing country i.e. a drug, device or surgery trial then you will need to register your study within the new UCL Interventional Clinical Trials Portal: www.ucl.ac.uk/ictm/portal by completing an Entry Questionnaire (EQ) and emailing a copy to ucl.trials.portal@ucl.ac.uk (cc.d to ethics@ucl.ac.uk). On receipt of your EQ, the ICTM Portal Review Group (who administer the portal) will discuss your EQ and an email response will be sent to you with information on the next steps in the process. General queries/questions regarding the Portal should be directed to: ucl.trials.portal@ucl.ac.ukStep 5
Disclosure and Barring Service (DBS) Checks (if required)
A criminal record check will be required by law if the research includes working in 'Regulated' activity with vulnerable groups as defined by the Safeguarding Vulnerable Groups Act 2006 or in a position of trust as defined by the Rehabilitation of Offenders Act Exception Order 1975. Further information and advice is available at the → Disclosure and Barring Service (DBS) Checks website. It is imperative that researchers to whom this applies take action promptly. It can take a month or more to arrange and complete the check; but it is important that researchers have received their clearance before entering a position of trust. Please quote your DBS Disclosure Number in section A2 of the application form. Current students or those who have applied for a place on a programme at UCL which needs DBS clearance must visit the Student Centre to have their DBS form checked and authorised, and should email studentrecords@ucl.ac.uk for information about this in the first instance.
Staff who need the DBS check as part of their work (for a research project, for example) should contact the Employment Contracts Administration of Human Resources at HR.ECA@ucl.ac.uk for a form to complete to decide what level of certificate is required. Once they have seen your completed form and made their decision, you will need to go to HR in person to collect the DBS form itself.
Step 6
Risk Assessment
In order to determine whether there are any risks associated with your research i.e. risks to yourself as the researcher and to those you are researching, it is important to carry out a risk assessment. It is a legal requirement that all research is assessed for risk. Refer to your Departmental arrangements for risk assessment procedures. Alternatively please refer to UCL Safety Services guidance on how to carry out a risk assessment. The guidance includes how to record the assessment which must be authorised by your Supervisor and retained for your records. Please e-mail: safety@ucl.ac.uk if you require further advice.
Step 7 (if required)
Insurance
The insurance for all UCL studies is provided by a commercial insurer. For the majority of studies the cover is automatic. However, staff or students undertaking the following types of studies will need to complete an insurance form (see → UCL Insurance) when applying to the UCL Research Ethics Committee for ethical approval of their project:
- intervention studies which enrol over 5,000 subjects
- all clinical intervention studies which enrol children aged 5 years and under where the aim is prophylaxis
- all clinical intervention studies where an inclusion criteria is pregnancy
- all intervention or clinical research studies conducted in the USA and Canada
Travel Insurance arrangements for students conducting research overseas an application form will need to be completed so that an insurance cover note can be issued. For further information and guidance please visit → Insurance website.
Step 8
Complete the application form and appendices
that are applicable to your study ensuring that both the Principal Researcher and the Head of your Department (OR Departmental Ethics Committee Chair/Departmental Ethics Lead) have signed the form.
Please find below sample copies of our ‘low’ and ‘high’ risk application forms and application guidance documents for review purposes only. Please do not attempt to complete these versions.
- Sample Low Risk Application Form
- Sample Guidelines for completing the Low Risk Application Form
- Sample High Risk Application Form
- Sample Guidelines for completing the High Risk Application Form
Recruitment Documents for Participants – All studies that involve the recruitment of participants will use recruitment documents such as information sheets and consent forms. See our ‘annotated’ template example forms: Template Consent Form and Template Participant Information Sheet. Also, see Advice on Formulating Participant Information Sheets.
Step 9
When the application is complete, please submit an authorised electronic copy of your application to: ethics@ucl.ac.uk.
It is important that your application is submitted as a single pdf document which contains the electronic signatures of both the Principal Researcher and Head of Department and includes any supporting documentation, all in one file. Please do not provide a scanned physically signed version as this creates a large pdf document.
Step 10
Following Approval
The Principal Researcher must report any proposed changes, any adverse events and if required report progress on an annual basis (see Key Responsibilities of the Principal Researcher following Approval).
 Close
Close